However, an increasing body of evidence suggests that XCI is incomplete with significant variability across tissues and even between the single cells 9, 10.  
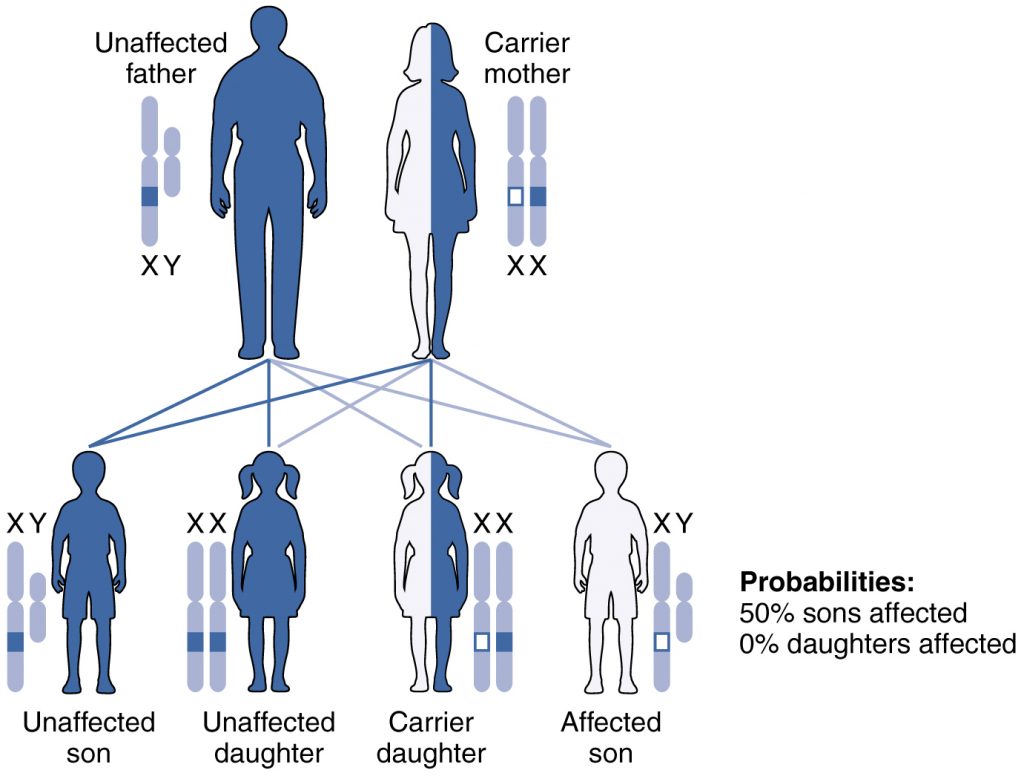
The inactive X chromosome can be observed as a tightly condensed heterochromatin body, also known as a Barr body 8. The process of XCI is initiated during early embryogenesis and is mediated via expression of the non-coding RNA X inactivation-specific transcript (XIST) from the X inactivation centre of the future inactive X chromosome 3, 4, 5, and a combination of epigenetic modifications including histone modifications and DNA methylation to achieve transcriptional silencing of the X-linked genes 6, 7. Notably, visible examples of diverse patterns of mosaicism in females heterozygous for easily distinguishable, monogenic, X-linked traits, such as X-linked fur colour in mice, were among the first lines of evidence for the hypothesis of random XCI 2. Due to random choice and the stability of the X chromosome inactivation (XCI) in mitotically derived cell lineages, the paternal or maternal alleles are expected to be mono-allelically expressed in different cell populations approximately 50% of the time, making females functionally haploid mosaics with respect to the X-linked genes. To balance allele dosage differences in X-linked genes between the sexes, dosage compensation (DC) mechanisms have evolved to randomly inactivate one of the X chromosomes in female cells 1, 2. In eutherian mammals, including humans, females inherit two copies of the X chromosome and males only one. Finally, we highlight examples of biologically relevant X-linked heterogeneity between the sexes that bias DC estimates if unaccounted for. Our DC estimates for complex traits and gene expression are consistent with a small proportion of genes escaping X-inactivation in a trait- and tissue-dependent manner. We find approximately twice as much X-linked genetic variation across the UK Biobank traits in males (mean h 2 SNP = 0.63%) compared to females (mean h 2 SNP = 0.30%), confirming the predicted DC effect. Here, we systematically examine the role of DC in humans in 20 complex traits in a sample of more than 450,000 individuals from the UK Biobank and 1600 gene expression traits from a sample of 2000 individuals as well as across-tissue gene expression from the GTEx resource. Quantitative genetics theory predicts that X-chromosome dosage compensation (DC) will have a detectable effect on the amount of genetic and therefore phenotypic trait variances at associated loci in males and females.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
